To operate the Blended Learning Center(BLC) at optimal level, maintenance will be performed every day at 8:30 AM and at 5:00 PM regularly which can take up to 30 minutes. Please consider scheduling your activity in the BLC platform accordingly.
Topic outline
- Introduction to pharmaceutical regulatory affairs

Introduction to pharmaceutical regulatory affairs
Regulatory affairs is a profession developed from the intention of
governments to protect public health by controlling the safety and efficacy of
products including pharmaceuticals, veterinary medicines, medical
devices, cosmetics so that that supplied products make a worthwhile contribution to public health and welfare.
Regulatory Affairs is a unique combination of science and management to achieve a commercially important goal within a drug-development organization.
The drug development process is a lengthy, complex and extremely costly albeit necessary process. Regulatory Affairs plays a crucial role in the pharmaceutical industry and is involved in all stages of drug development and also after drug approval and marketing.
Regulatory Affairs is an attractive career choice for graduate students from a scientific background who enjoy communication and team work, are comfortable with multi-tasking and are eager to expand their knowledge in the wide realms of the Pharmaceutical world.
It gives strategic and technical advice at the highest level in their companies, right from the beginning of the development of a product, making an important contribution both commercially and scientifically to the success of a development program and the company as a whole.
- Course Information:
Course Information:
Course Information:
Name of the course: Pharmaceutical Regulatory Affairs
Course Code: BPH 425
Total credits: 2
Program: B. Pharm.
Course Instructor:

Mr. Md. Anamul Haque
Assistant Professor
Department of Pharmacy
ID: 710002372
Email: anamul.ph@diu.edu.bd
Cell Phone:01767-290194
Faculty of Allied Health Sciences
Daffodil International University
Dhaka, Bangladesh

- General Instruction/Guidelines for students:

General Instruction/Guidelines for students:

- All the students registered for this course have to enroll in Moodle.
- Students can find all the course materials from Moodle.
- All the students have to submit the soft copy of their "Assignment" in Moodle under assignment section created here and for this they will be graded here.
- One discussion or feedback forum is created under each of the lecture Students have to give their feedback on these forum and marks will be given for their feedback
- Any announcement regarding the class will be posted on Moodle. So they have to keep themselves always active on Moodle.
- All the quizzes and presentation will be held on online (Moodle) and may be few of the class will be held on face to face class and it will be announced before the class.
- The question pattern and the syllabus for the quizzes, midterm and final exam is given here under each of the section (quizzes, midterm and final).
- There are Midterm and Final Examination presentation forum under these sections where students can discuss with each other about their midterm and final exam syllabus, any problem regarding the exam etc.
- Course details

Course details
Rationale:
This subject deals with the registration process of drugs and drugs
related materials.Licensing procedure for a pharmaceutical company and product as well.
Course Learning Outcomes:
- Explain basic requirements for drug registration.
-
Illustrate different
Stage and critical parameters of registration.
- Keeping track of the ever-changing
legislation in all the regions in which a company wishes to distribute its
products.
- Advising on legal
and scientific restraints and requirements
Course Contents:
1. Introduction to regulatory affairs and its scope.
2. Drug controlling authority of
Bangladesh and pharmacy council.
3. International regulatory agencies.
4. QA and regulatory requirements in pharmaceuticals
5. Acts, ordinance, policies regarding drug control in Bangladesh
6. Approval procedure for new pharmaceuticals in Bangladesh
7. Code of ethics for pharmacists.
Total marks: 100 Total credits: 3 |
Breakdown of marks |
Assessment types | Number of assessments | Marks in each assessment | Total marks |
Class test | 3 class tests | 10 marks | 10 marks (Average) |
Assignment | 1 assignment | 5 marks | 5 marks |
Presentation | 1 presentation | 5 marks | 5 marks |
Attendance | Base on class attendance | 5 marks |
Midterm exam | 25 marks (Question types: MCQ,T/F,SQ) | 25 marks |
Final exam | In two slots(Question types: MCQ,T/F,SQ) | 40 marks |
Online activity | Case study, viva, interactive session etc. | 10 marks |
Total marks: | 100 |
- .1. Introduction to regulatory affairs and its scope.
.1. Introduction to regulatory affairs and its scope.
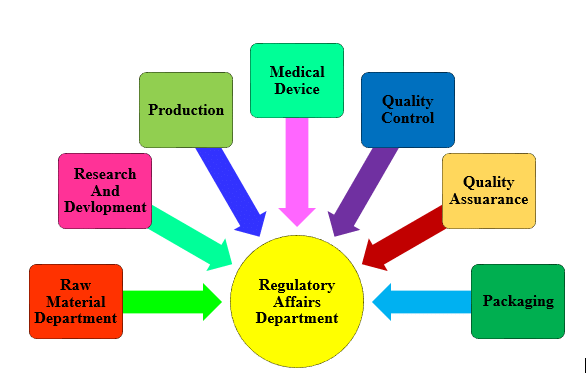
Learning Outcomes:
- Able to get general idea about regulatory affairs.
- Understand scope and areas of RA.
- Figure out the goal of regulatory affairs.
- Role of RAD in a pharmaceuticals.
Course content:
- Definition, scope & Goals for Regulatory Affairs.
- Contribution of RAD in a pharmaceuticals.
- Function of regulatory professionals.
- Question & Answer Forum (Drug regulatory affairs intro)
Question & Answer Forum (Drug regulatory affairs intro)
- 2. Drug controlling authority of Bangladesh and pharmacy council.
2. Drug controlling authority of Bangladesh and pharmacy council.


Learning Outcomes:
- Able to get general idea about drug regulatory authority of Bangladesh.
- Understand scope and areas of DGDA
- Get idea about organogram of DGDA
- Figure out Scope and functions of Pharmacy Council of Bangladesh.
Course content:
- Functions
and activities of DGDA,
- Drug Control committee (DCC) and related technical committees.
- Ordinance, different Committees
- Scope and functions of Pharmacy Council of Bangladesh.
- Assignment
- 3. International drug regulatory agencies
3. International drug regulatory agencies

Learning Outcomes:
- Get general idea about International drug regulatory agencies.
- Learn about cGMP, ICH guidelines.
- Know the role and scope of FDA, MHRA,TGA.
- Get into know about ISO:9001
Course content:
- Introduction to international drug regulatory agencies.
- FDA, MHRA, TGA, ANVISA, GCC
- QUIZ 1
- Mid term examination
Mid term examination

Mid-Term Exam:
Total Marks: 15
Total Time: 17 minutes
Chapter :
1. Introduction to RA
2. Drugs controlling authority in Bangladesh
3. International drug regulatory agencies.
Assessment strategy:
MCQ: 10 marks
True/False: 5 marks
Level 1: Ten questions relating to subjects topic and general idea.
Level 2: Four questions to judge understand ability of particular topics.
Level 3: Four questions relating to elucidation of some pharmacological facts.
Level 4: Two questions to assess students critical thinking.
- 4. QA and regulatory requirements in pharmaceuticals
4. QA and regulatory requirements in pharmaceuticals

Learning Outcomes:
- Learn about quality management system.
- Know the components of QA
- Will be introduced about importance of QA
Course content:
- Introduction to total quality management system.
- Concept of QA and regulatory documents.
- Importance of stability study.
- Scope and function of QA.
- QUIZ 2
- 5. Acts, rules, ordinance and laws regarding regulation of drugs in Bangladesh.
5. Acts, rules, ordinance and laws regarding regulation of drugs in Bangladesh.

Learning Outcomes:
- Know about different laws.
- Get idea about acts regarding drugs regulation.
Course content:
- Pharmacy Legislation Drug Act 1940
- The Narcotics
(Control) Act 1990
- National Drug Policy 2016
- 6. Approval process, format and registration of pharmaceuticals in Bangladesh
6. Approval process, format and registration of pharmaceuticals in Bangladesh

Learning Outcomes:
- Able to get general idea about pharmaceuticals registration procedure.
- Get idea about product registration formalities.
- Figure out the required documents for a new pharmaceutical company.
- How to conduct a formal inspection and documentation.
Course content:
- Licensing procedure for a pharmaceutical company
- Documentation for a new pharmaceuticals.
- Role of drug control committee (DCC).
- Recipe approval & pricing.
- QUIZ 3
- Presentation
Presentation
|
Topics
|
|
FDA
|
|
MHRA
|
|
TGA
|
|
ICH
|
|
Dossier
|
|
Drug Master File
|
|
CTD
|
|
DGDA
|
|
QAD in pharmaceuticals
|
|
Stability study
|
|
QCD
|
|
GMP
|
|
Drug policy 1982
|
|
Drug policy 2016
|
Share the presentation video link of your assigned topics from the above by 25.11.2020
- Final examination
Final examination

Final Exam Fall-2020
Total marks: 50
Assessment strategy:
Online Exam at BLC: 30 Marks Time 40 minutes
MCQ 20 marks, True/False 10 marks
Oral Assessment 15 Marks
Online activity 5 marks
Syllabus
Quality management system in pharmaceuticals
Act, laws and drug policies, Drug ordinance 1982
Approval process of pharmaceutical in Bangladesh
Question types
Level 1: 15 questions relating to subjects topic and general idea.
Level 2: 8 questions to judge understand ability of particular topics.
Level 3: 4 questions relating to elucidation of some pharmacological facts.
Level 4: 3 question to assess students critical thinking.
- Lesson of life
- Compliments